All corrosion and scale formation in water chemistry is controlled by the water’s
pH Alkalinity CO₂
Water hardness (grains of hardness) – The level of dissolved solids in the water
Alkalinity – The capacity of water to resist acidification (the process of something becoming acidic)
pH – A measure of acidity and alkalinity of a solution
CO₂ (Carbon dioxide) – Determines pH of water. The amount of CO₂ in a solution also determines the pH of the water. The most common source of acidity in water is dissolved CO₂. The more CO₂ in the water, the lower the pH. This is because CO₂ comes aqueous in water, a small portion of it becomes carbonic acid solution.
20th Century Approach
Once upon a time . . . . . .
Some companies and products once upon a time were so large and well-established, it seems like they could never go out of business. But innovation can quickly leave a business behind if it can’t adjust to new ways of operating, or to changing customer demand for new products and services.


You can’t take on 21st century tasks with 20th century tools and hope to get the job done
Cathy N. Davidson
Professor – Duke University








Phosphates
1887: First recognized in water treatment.
1930’s: “Threshold Treatment”- use of few ppm sodium hexa-metaphosphate powder to potable water for control of calcium carbonate scale.
1970’s: Zinc phosphates introduced for the treatment of low hardness aggressive water supplies
(Murray AWWA 1969)
Not effective when water is over 10 grains
Increase bacterial growth in plumbing systems (RAISE pH)
Less effective with time; reverts from poly to orthophosphates
Increases TDS
Capacity drops as grains of hardness go up
Always being released from the cartridges. Polyphosphates continues to be elevated when equipment is not in use thus causing huge pH swings, which makes the water taste metallic.
Polyphosphates are additives that are dosed into water at a controlled rate to inhibit scale. The dosing can be performed manually or automatically at levels of around two parts per million (ppm). Water filters that contain polyphosphates usually hold them within the cartridge and slowly dose the water as it passes through.
The scale-inhibition technique works by joining the magnesium and calcium ions to a phosphate ion . This helps prevent them from accumulating on heating coils and blocking water lines. Dosing does not physically remove the calcium and magnesium from the water, but instead simply inhibits their ability to form.
Polyphosphate treatment works best for soft to moderately hard water (=< 7 grains) for applications such as ice or coffee but becomes less effective in very hard water areas and for applications such as espresso or steam where other technologies such as ion-exchange or reverse osmosis will be required.
Reason to replace your polyphosphates filter:
Over time, as polyphosphates get added and mixed into the water they diminish. (Think of it like eating a piece of hard candy – it gets smaller and smaller until it’s fully dissolved).
When exhausted, there’s no more protection against scale.₁
https://www.3m.com/3M/en_US/water-quality-us/applications/commercial-food-service-water-filtration/foodservice-faqs/

Ion Exchange
1903: Invented
Exchanging Calcium and Magnesium ions for Sodium and Hydrogen Ions
Sodium and Hydrogen ions linked to high blood pressure and acidic body cells that can lead to cancer
Increases TDS
Bypass needed (NOT 100% FILTRATION) for espresso & coffee (good extraction)
Taste, expensive and environmental issues
Capacity and size
Lowers hardness to help reduce scale
Adds 0.79 ppm of carbon dioxide (CO₂) for each ppm of sodium bicarbonate (HCO₃) . Steam is condensed, carbon dioxide (CO₂) dissolves in water (Lower pH) + increases Hydrogen ion (NaHCo₃) concentration (Lower alkalinity)
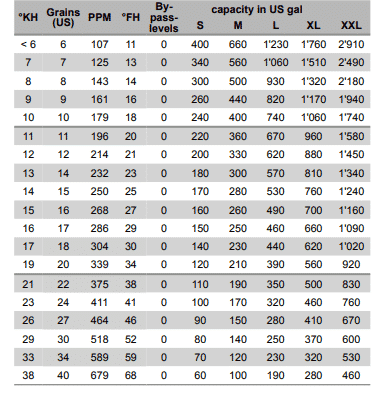
Most ion exchange filters are rated for 7 – 12 grains of hardness
The table below illustrates how water hardness decreases capacity

THE TRADE OFF
Addressing one issue scale (temporary hardness) but creating scale (permanent hardness) and /or corrosion
Biofouling
Biofouling
The accumulation of microorganisms, plants, algae, or small animals on wetted surfaces that have a mechanical function, causing structural or other functional deficiencies.
Calcium phosphates provides nutrients for bacteria (slime growth) to thrive on
Reverse Osmosis
Space: Need an area to accommodate the storage tank
Time: Lengthy process to produce (1) usable gallon of water
Waste: Use 2 – 3 gallons of water to create (1) gallon of RO water (increase costs due to unused water)
Corrosion: Plastic tubing and parts are used to avoid leeching as the water is acidic
TDS: Some level is needed to the equipment to function properly
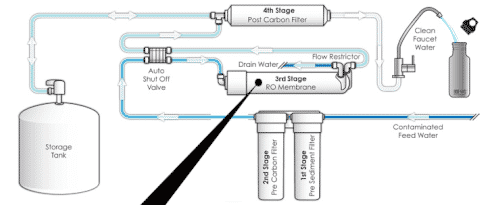
Using a reverse osmosis system to treat hard water (above 7 grains) shortens the life of the RO membrane. This will lead to more frequent membrane replacements.
The prefilters and post-filters should be changed every 6 months to 1 year. Depending on your water conditions, the RO membrane should be replaced every 1-2 years
21st Century Technology – TAC and NAC



1st generation
Template Assisted Crystallization (TAC)
Instead of removing the molecules that cause hard water these hard water mineral molecules into converted to low energy ions process called Template Assisted Crystallization (TAC), which prevents molecules from sticking or calcifying. The media is comprised of calcite crystals that provide template sites that cause dissolved calcium, magnesium and bicarbonate to combine into microscopic crystals. Once formed, these crystals detach from the media, and flow freely through the system.
Due the slower timing process, over time the TAC media will start to break down, which is typically seen in the 1st year after installation. TAC media is also not recommended for temperatures higher than 140°. CO2 blends into water. TAC lowers pH and in most cases water becomes acidic. Removes existing scale but has no corrosion protection and can make o-rings and gaskets leak.
Water treatment comparison

3rd generation
Nucleation Assisted Crystallization (NAC)
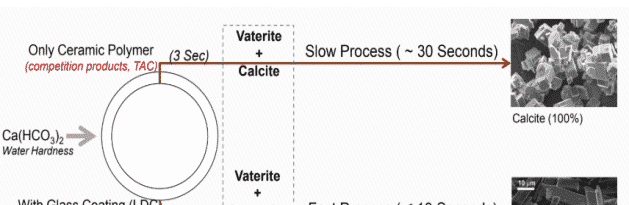
“SP (Scale Prevention) 3” indicates a unique transformation of water hardness Ca(HCO₃)₂ into (3) components:
CaCO₃ (micro-crystals) Aragonite 2. CO₂ (colloid)
H₂O (pure water)
Phase 1: Filtersorb SP3 double ceramic beads accelerates the transformation of the calcium and magnesium minerals out of the water solution into harmless nano sized aragonite crystal particles. Aragonite exists as tiny, crystals that do not cling to surfaces. Its layered double coated (LDC) catalytic agent ensures the Calcite morphs into aragonite crystals. This is currently the only media available causing this transformation.
Phase 2: As the minerals flow through the piping systems and equipment, the aragonite crystals dissolve additional calcium removing any pre-existing calcium deposits.
Phase 3: After the old calcium deposit are removed, Filtersorb SP3 form a smooth 3 – 5 micro thick corrosion prevention layer on existing pipes, o-rings, gaskets, and equipment.
No TDS change: No elements of the water are removed. No ion-exchange chemistry therefore the TDS stays the same before and after treatment.
No pH change: pH of the water remains same. Making the treated water suitable for almost any use for corrosion.
This faster process of the molecules, NAC life expectancy is typically (3) years and can sustain temperatures of up to 189°
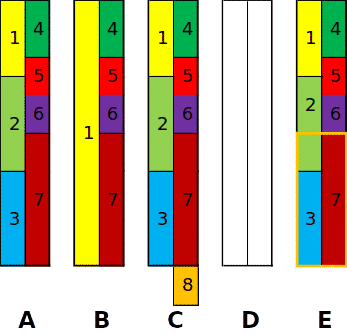
A – The natural state of water
High Magnesium (Mg) and Calcium (Ca) + Low Sodium (Na) and Potassium (K)
B – After ion exchange (water softener) treatment
Increased Sodium (Na) + increased Chloride (Cl) = CORROSION
Increased Sodium (Na) + Bicarbonate (HCO₃) = SCALE
C – After Phosphate treatment
Increased Sulfate (SO₄) + increased Silica (SiO₂) = CORROSION
Addition of Phosphates (PO₄³-) = SCALE (PERMENANT)
D – After Reverse Osmosis treatment (non-blended / not remineralized)
Removal of all Cations and Anions + CO₂ (from the air) = CORROSION
E – After Filtersorb SP3 or CT treatment
Encapsulation treatment protects against corrosion & scale while retaining water’s natural state
