Chloramines and Chlorides
Chloramines: A disinfectant used by municipalities for water treatment
Chlorides: Natural salt and / or salt by products created by people (snow removal, water softeners)
Both can do serious damage to plumbing an kitchen equipment. Especially when combined with high temperatures.



Corrosive Effect of Chloramine on Water Heater Pipes
First photo: corrosion on top of water heater caused by leak
Second photo: hot water output pipe
Third photo: cold water intake pipe
Note that heated chloraminated water is much more corrosive than cold chloraminated water.
Second photo: hot water output pipe
Third photo: cold water intake pipe
Note that heated chloraminated water is much more corrosive than cold chloraminated water.
Chlorides
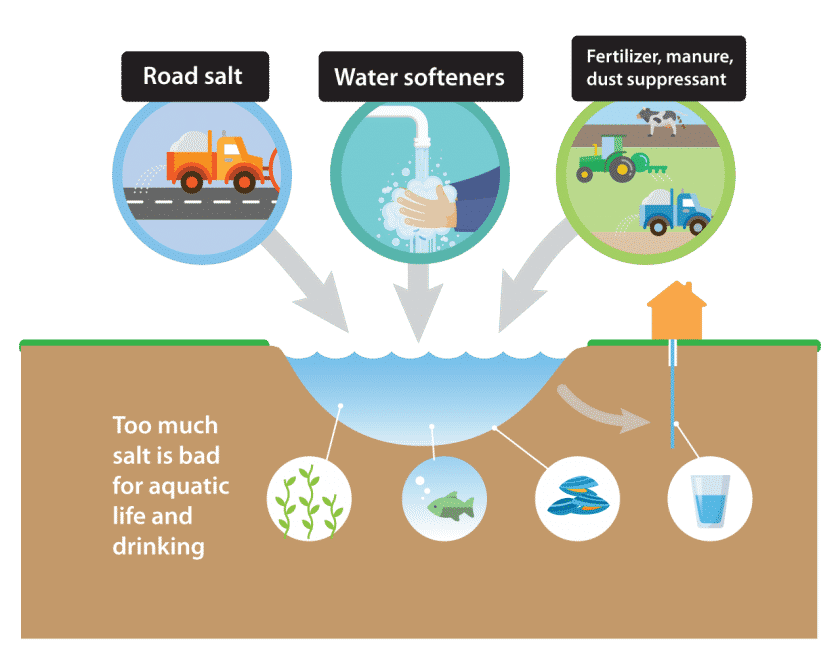
Chloride from both de-icing salt and water softener salt gets into lakes and streams, and groundwater that supplies drinking water.
It takes only one teaspoon of salt to permanently pollute five gallons of water.
Once in the water, there is no easy way to remove the chloride.
https://www.pca.state.mn.us/water/chloride-101
